Our Work:
|
|
5. Shu-Bo Hu, Xiao-Yong Zhai, Hong-Qiang Shen and Yong-Gui Zhou.* Iridium-catalyzed Asymmetric Hydrogenation of Polycyclic Pyrrolo/Indolo[1,2-a]quinoxalines and Phenanthridines. Adv. Synth. Catal. 2018, 360, 1334-1339. |

|
|
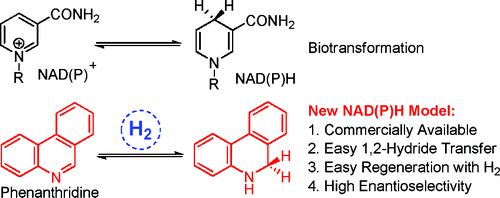
4. Qing-An Chen, Kai Gao, Ying Duan, Zhi-Shi Ye, Lei Shi, Yan Yang and Yong-Gui Zhou.* Dihydro phenanthridine: A New and Easily Regenerable NAD(P)H Model for Biomimetic Asymmetric Hydrogenation. J. Am. Chem. Soc. 2012, 134, 2442-2448. |
|
|
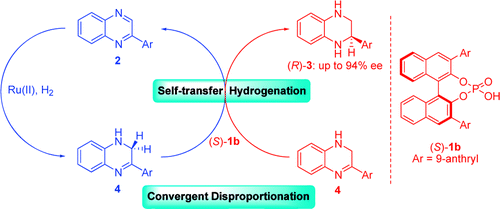
3. Qing-An Chen, Duo-Sheng Wang, Yong-Gui Zhou,* Ying Duan, Hong-Jun Fan,* Yan Yang, and Zhang Zhang. Convergent Asymmetric Disproportionation Reactions: Metal/Bronsted Acid Relay Catalysis for Enantioselective Reduction of Quinoxalines. J. Am. Chem. Soc. 2011, 133, 6126-6129. |
|
|
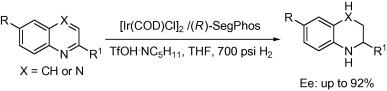
2. Duo-Sheng Wang, Yong-Gui Zhou.* Asymmetric hydrogenation of quinolines activated by Bronsted acids. Tetrahedron Lett. 2010, 51, 3014-3017. |
|
|
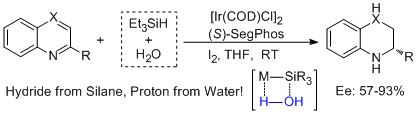
1. Da-Wei Wang, Duo-Sheng Wang, Qing-An Chen, and Yong-Gui Zhou.* Asymmetric Hydrogenation with Water/Silane as the Hydrogen Source. Chem. Eur. J. 2010, 16, 1133-1136. |
Others' Work:
|
|
16. Mu-Wang Chen, Zhihong Deng, Qin Yang, Jian Huang and Yiyuan Peng.* Enantioselective Synthesis of Trifluoromethylated Dihydroquinoxalinones via Palladium-catalyzed Hydrogenation. Org. Chem. Front. 2019, 6, 746-750. |

|
|
15. Songlei Li, Wei Meng, and Haifeng Du.* Asymmetric Transfer Hydrogenations of 2,3-Disubstituted Quinoxalines with Ammonia Borane. Org. Lett. 2017, 19, 2604-2606. |
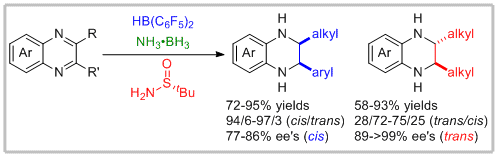
|
|
14. Kosuke Higashida,a Haruki Nagae,a and Kazushi Mashimaa.* Iridium-Catalyzed Asymmetric Hydrogenation of Tosylamido-Substituted Pyrazines for Constructing Chiral Tetrahydro-pyrazineswith an Amidine Skelton. Adv. Synth. Catal. 2016, 358, 3949-3954. |
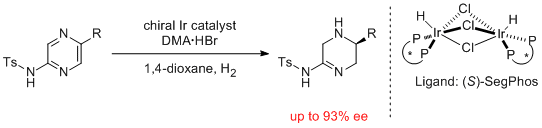
|
|
13. Noriyoshi Arai, Yu Saruwatari, Kotaro Isobe, and Takeshi Ohkumaa.* Asymmetric Hydrogenation of Quinoxalines, Benzoxazines, and a Benzothiazine Catalyzed by Chiral Ruthenabicyclic Complexes. Adv. Synth. Catal. 2013, 355, 2769-2774. |

|
|
12. Steffen Fleischer, Shaolin Zhou, Svenja Werkmeister, Kathrin Junge, and Matthias Beller.* Cooperative Iron–Bronsted Acid Catalysis: Enantioselective Hydrogenation of Quinoxalines and 2H-1,4-Benzoxazines. Chem. Eur. J. 2013, 19, 4997-5003. |
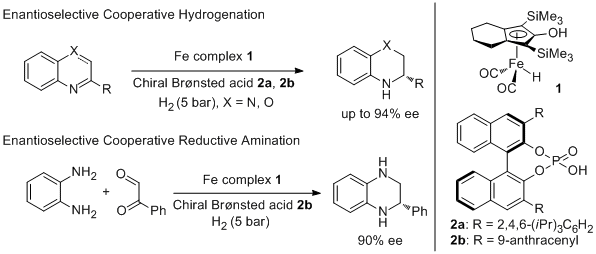
|
|
11. Takuto Nagano, Atsuhiro Iimuro, Rino Schwenk, Takashi Ohshima, Yusuke Kita, Antonio Togni and Kazushi Mashima.* Additive Effects of Amines on Asymmetric Hydrogenation of Quinoxalines Catalyzed by Chiral Iridium Complexes. Chem. Eur. J. 2012, 18, 11578-11592. |
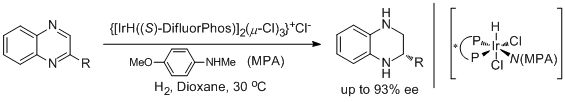
|
|
10. Damien Cartigny, Farouk Berhal, Takuto Nagano, Phannarath Phansavath, Tahar Ayad, Jean-Pierre Genêt, Takashi Ohshima, Kazushi Mashima, and Virginie Ratovelomanana-Vidal.* General Asymmetric Hydrogenation of 2-Alkyl- and 2-Aryl-Substituted Quinoxaline Derivatives Catalyzed by Iridium-Difluorphos: Unusual Halide Effect and Synthetic Application. J. Org. Chem. 2012, 77, 4544-4556. |
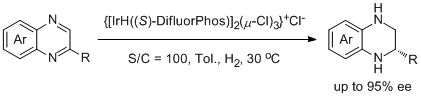
|
|
9. Jie Qin, Fei Chen, Ziyuan Ding, Yan-Mei He, Lijin Xu, and Qing-Hua Fan.* Asymmetric Hydrogenation of 2- and 2,3-substituted Quinoxalines with Chiral Cationic Ruthenium Diamine Catalysts. Org. Lett. 2011, 13, 6568-6571. |
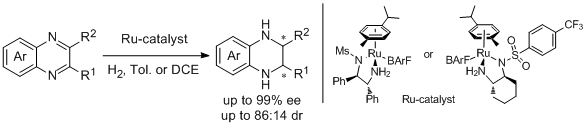
|
|
8. Damien Cartigny, Takuto Nagano, Tahar Ayad, Jean-Pierre Genet, Takashi Ohshim,* Kazushi Mashima* and Virginie Ratovelomanana-Vidal.* Iridium-Difluorphos-Catalyzed Asymmetric Hydrogenation of 2-Alkyl- and 2-Aryl-Substituted Quinoxalines: A General and Efficient Route into Tetrahydroquinoxalines. Adv. Synth. Catal. 2010, 352, 1886-1891. |

|
|
7. Magnus Rueping,* Francisco Tato, and Fenja. R. Schoepke. The First General, Efficient and Highly Enantioselective Reduction of Quinoxalines and Quinoxalinones. Chem. Eur. J. 2010, 16, 2688-2691. |
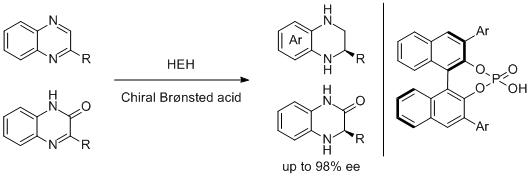
|
|
6. Nataša Mršić, Thomas Jerphagnon, Adriaan J. Minnaard,* Ben L. Feringa,* and Johannes G. de Vriesa.* Asymmetric Hydrogenation of Quinoxalines Catalyzed by Iridium/PipPhos. Adv. Synth. Catal. 2009, 351, 2549-2552. |
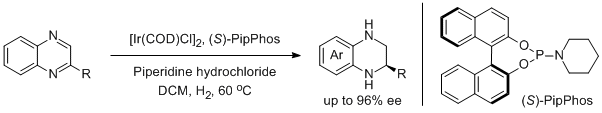
|
|
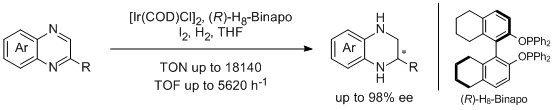
5. Weijun Tang, Lijin Xu,* Qing-Hua Fan,* Jun Wang, Baomin Fan, Zhongyuan Zhou, Kim-hung Lam, and Albert S. C. Chan.* Asymmetric Hydrogenation of Quinoxalines with Diphosphinite Ligands: A Practical Synthesis of Enantioenriched, Substituted Tetrahydroquinoxalines. Angew. Chem. Int. Ed. 2009, 48, 9135-9138. |
|
|
4. Julian P. Henschke,* Mark J. Burk, Christophe G. Malan, Daniela Herzberg, Justine A. Peterson, Andrew J. Wildsmith, Christopher J. Cobley, Guy Casy.* Synthesis and Applications of HexaPHEMP, a Novel Biaryl Diphosphine Ligand. Adv. Synth. Catal. 2003, 345, 300-307. |

|
|
3. Christopher J. Cobley, Julian P. Henschke. Enantioselective Hydrogenation of Imines Using a Diverse Library of Ruthenium Dichloride(diphosphine)(diamine) Precatalysts. Adv. Synth. Catal. 2003, 345, 195-201. |
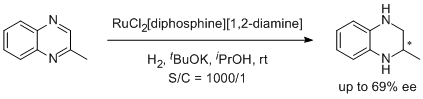
|
|
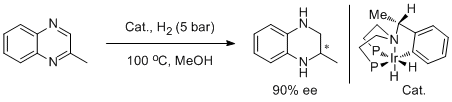
2. Claudio Bianchini,* Pierluigi Barbaro, Giancarlo Scapacci, Erica Farnetti, and Mauro Graziani. Enantio- selective Hydrogenation of 2-Methylquinoxaline to (-)-(2S)-2-Methyl-1,2,3,4-tetrahydroquinoxaline by Iridium Catalysis. Organometallics 1998, 17, 3308-3310. |
|
|
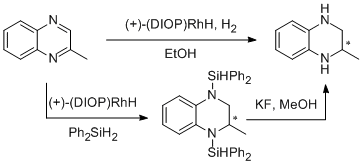
1. Shizuaki Murata*, Takashi Sugimoto, and Sadao Matsuura, Hydrogenation and Hydrosilylation of Quinoxaline by Homogeneous Rhodium Catalysts. Heterocycles. 1987, 26, 763-766.
|
|