Our Work:
|
|
21. Mu-Wang Chen, Xian-Feng Cai, Zhang-Pei Chen, Lei Shi and Yong-Gui Zhou.* Facile Construction of Three Contiguous Stereogenic CentersviaDynamic Kinetic Resolution in Asymmetric Transfer Hydrogenation of Quinolines. Chem. Commun. 2014, 50, 12526-12529. |

|
|
20. Xian-Feng Cai, Wen-Xue Huang, Zhang-Pei Chen and Yong-Gui Zhou.* Palladium-Catalyzed Asymmetric Hydrogenation of 3-Phthalimido Substituted Quinolines. Chem. Commun. 2014, 50, 9588-9590. |
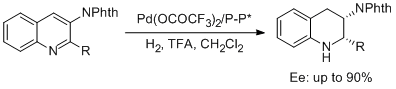
|
|
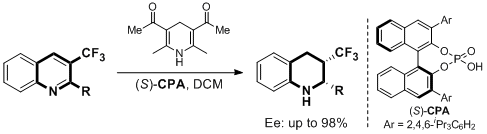
19. Ran-Ning Guo, Zhang-Pei Chen, Xian-Feng Cai and Yong-Gui Zhou.* Asymmetric Transfer Hydroge-nation of 3-(Trifluoromethyl)quinolines. Synthesis 2014, 46, 2751-2756. |
|
|
18. Xian-Feng Cai, Ran-Ning Guo, Guang-Shou Feng, Bo Wu and Yong-Gui Zhou.* Chiral Phosphoric Acid-Catalyzed Asymmetric Transfer Hydrogenation of Quinolin-3-amines. Org. Lett. 2014, 16, 2680-2683. |

|
|
17. Xian-Feng Cai, Ran-Ning Guo, Mu-Wang Chen, Lei Shi and Yong-Gui Zhou.* Synthesis of Chiral Exocyclic Amines via Asymmetric Hydrogenation of Aromatic Quinolin-3-amines. Chem. Eur. J. 2014, 20, 7245-7248. |

|
|
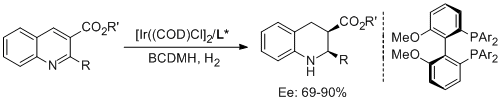
16. Zhang-Pei Chen, Zhi-Shi Ye, Mu-Wang Chen and Yong-Gui Zhou.* Enantioselective Synthesis of Endo-CyclicBeta-Amino Acids with Two Contiguous Stereocenters via Hydrogenation of 3-Carbalkoxy-2-Substituted Quinolines.Synthesis 2013, 3239-3244. |
|
|
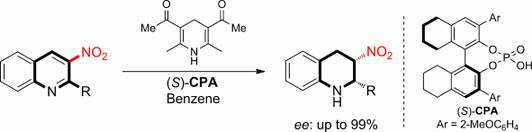
15. Xian-Feng Cai, Mu-Wang Chen, Zhi-Shi Ye, Ran-Ning Guo, Lei Shi, Yan-Qin Li and Yong-Gui Zhou.* Asymmetric Transfer Hydrogenation of Aromatic Nitro Compounds, 3-Nitroquinolines: A Facile Access to Cyclic Nitro Compounds with Two Contiguous Stereocenters. Chem. Asian J. 2013, 8, 1381-1385. |
|
|
14. De-Yang Zhang, Duo-Sheng Wang, Min-Can Wang,* Chang-Bin Yu, Kai Gao and Yong-Gui Zhou.*Synthesis of Electronically Deficient Atropisomeric Bisphosphine Ligands and Their Application in Asymmetric Hydrogenation of Quinolines. Synthesis. 2011, 2796-2802. |
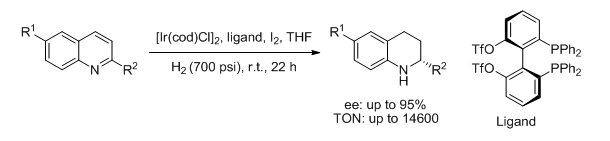
|
|
13. Jie Tang, Guo-Fang Jiang,* andYong-Gui Zhou.* Convenient Synthesis of Optically Pure 8-Methoxy-2-methyl-1,2,3,4-tetrahydroquinoline and 2-Methyl-1,2,3,4-tetrahydroquinoline. Heterocycles. 2010,82,887-893. |
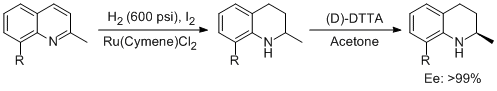
|
|
12. Da-Wei Wang, Duo-Sheng Wang, Qing-An Chen,Yong-Gui Zhou.* Asymmetric Hydrogenation with Water/Silane as the Hydrogen Source. Chem. Eur. J. 2010, 16, 1133-1136. |
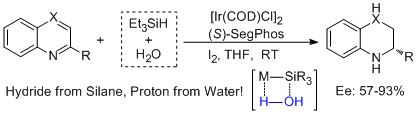
|
|
11. Duo-Sheng Wang, Juan Zhou, Da-Wei Wang, Yin-Long Guo* and Yong-Gui Zhou.* Inhibiting Deactivation of Iridium Catalysts with Bulky Substituents on Coordination Atoms. Tetrahedron Lett. 2010, 51, 525-528. |
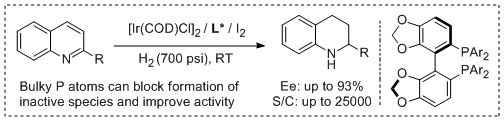
|
|
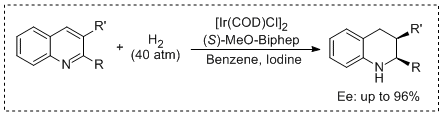
10. Da-Wei Wang, Xiao-Bing Wang, Duo-Sheng Wang, Sheng-Mei Lu, Chang-Bin Yu, Yong-Gui Zhou.*Highly Enantioselective Iridium-Catalyzed Hydrogenation of 2-Benzylquinolines, 2-Functionalized and 2,3-Disubstituted Quinolines. J. Org. Chem. 2009, 74, 2780-2787. <Highlighted by SYNFACTS 2009, 770.> |
|
|
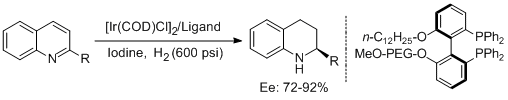
9. Xiao-Bing Wang and Yong-Gui Zhou.* Synthesis of Tunable Bisphosphine Ligands and Their Application in Asymmetric Hydrogenation of Quinolines. J. Org. Chem. 2008, 73, 5640-5642. |
|
|
8. Sheng-Mei Lu, Xiu-Wen Han, Yong-Gui Zhou.* An Efficient Catalytic System for the Hydrogenation of Quinolines. J. Organomet. Chem. 2007, 692, 3065-3069. |
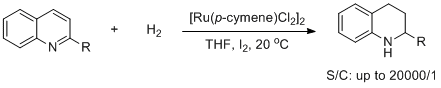
|
|
7. Da-Wei Wang, Wei Zeng, Yong-Gui Zhou.* Iridium-catalyzed Asymmetric Transfer Hydrogenation of Quinolines with Hantzsch Esters.Tetrahedron: Asymmetry 2007, 18, 1103-1107. <Highlighted by SYNFACTS 2007, 830.>
|
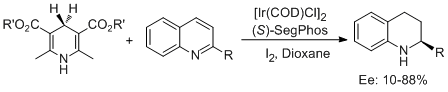
|
|
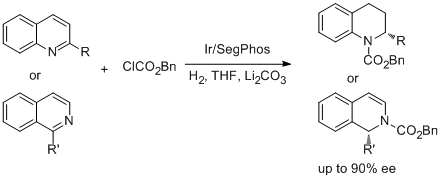
6. Sheng-Mei Lu, You-Qing Wang, Xiu-Wen Han and Yong-Gui Zhou.* Asymmetric Hydrogenation of Quinoline and Isoquinolines Acitivated by Chloroformates. Angew. Chem. Int. Ed. 2006, 45, 2260-2263. <Highlighted by SYNFACTS 2006, 582.> <Highlighted by Angew. Chem. Int. Ed. 2013,52, 9616>
|
|
|
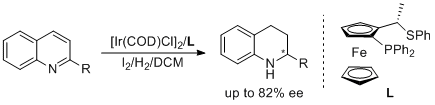
5. Yu-Jun Zhao, You-Qing Wang, Yong-Gui Zhou.* Application of Ferrocene-Derived Chiral S,P-Donor Ligands for Iridium-catalyzed Asymmetric Hydrogenation of Quinolines. Chin. J. Catal. 2005, 26, 737-739. |
|
|
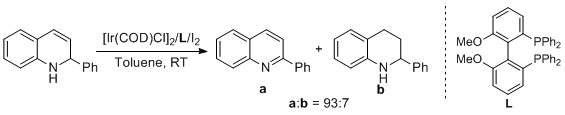
4. Sheng-Mei Lu, You-Qing Wang, Xiu-Wen Han, Yong-Gui Zhou.* Homogeneous Iridium-catalyzed Dehydro-aromatization of 2-Substituted-1,2-dihydro-Quinolines. Chin. J. Catal. 2005, 26, 287-290. |
|
|
3. Sheng-Mei Lu, Xiu-Wen Han, Yong-Gui Zhou.* Asymmetric Hydrogenation of Quinolines Catalyzed by Iridium with Chiral Ferrocenyloxazoline Derived N,P Ligands. Adv. Synth. Catal. 2004, 346, 909-912. |

|
|
2. Peng-Yu Yang, Yong-Gui Zhou.* The Enantioselective Total Synthesis of Alkaloid (-)-Galipeine.Tetrahedron: Asymmetry 2004, 15, 1145-1149. |

|
|
1. Wen-Bo Wang, Sheng-Mei Lu, Peng-Yu Yang, Xiu-Wen Han, Yong-Gui Zhou.* Highly Enantioselective Iridium-Catalyzed Hydrogenation of Heteroaromatic Compounds, Quinolines. J. Am. Chem. Soc. 2003, 125, 10536-10537. |

Others' Work:
|
|
60. Siyuan Sun and Pavel Nagorny.* Exploration of Chiral Diastereomeric Spiroketal (SPIROL)-Based Phosphinite Ligands in Asymmetric Hydrogenation of Heterocycles. Chem. Commun. 2020, 56, 8432-8435.  ; ;
|

|
|
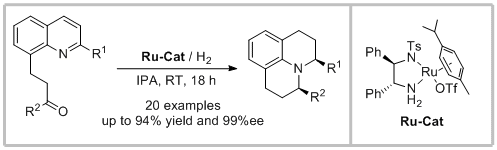
59. Li-Ren Wang, Dan Chang, Yu Feng,* Yan-Mei He, Guo-Jun Deng* and Qing-Hua Fan.* Highly Enantioselective Ruthenium-Catalyzed Cascade Double Reduction Strategy: Construction of Structurally Diverse Julolidines and Their Analogues. Org. Lett. 2020, 22, 2251-2255.
|
|
|
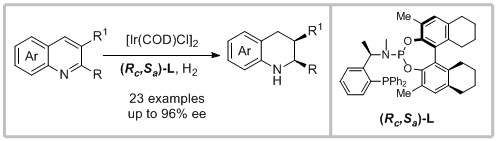
58. Xin-Hu Hu and Xiang-Ping Hu.* Highly Diastereo- and Enantioselective Ir-Catalyzed Hydrogenation of 2,3-Disubstituted Quinolines with Structurally Fine-Tuned Phosphine-Phosphoramidite Ligands. Org. Lett. 2019, 21, 10003-10006.
|
|
|
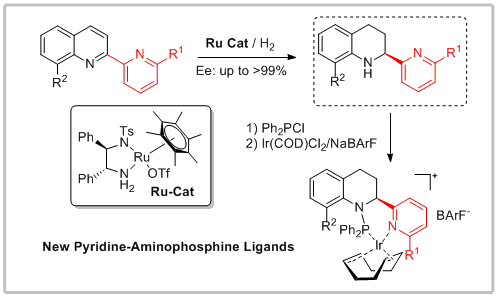
57. Youran Liu, Fei Chen, Yan-Mei He, Chenghao Li and Qing-Hua Fan* Enantioselective Synthesis of Tunable Chiral Pyridine–Aminophosphine Ligands and Their Applications in Asymmetric Hydrogenation. Org. Biomol. Chem. 17, 5099-5105.
|
|
|
56. Ya Chen, Yixiao Pan, Yan-Mei He* and Qing-Hua Fan.* Rapid Consecutive Intermolecular Reductive Amination/Asymmetric Hydrogenation: Facile Access to Sterically Tunable Chiral Vicinal Diamines and N-Heterocyclic Carbenes. Angew. Chem. Int. Ed. 2019, 58, 16831-16834.
|

|
|
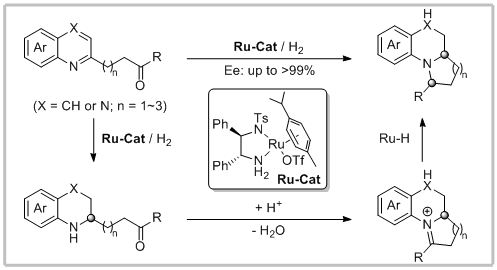
55. Ya Chen, Yan-Mei He, Shanshan Zhang, Tingting Miao and Qing-Hua Fan.* Rapid Construction of Structurally Diverse Quinolizidines, Indolizidines, and Their Analogues via Ruthenium-Catalyzed Asymmetric Cascade Hydrogenation/Reductive Amination. Angew. Chem. Int. Ed. 2019, 58, 3809-3813.
|
|
|
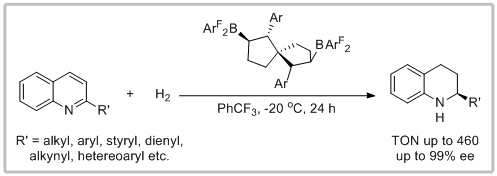
54. Xiang Li, Jun-Jie Tian, Ning Liu, Xian-Shuang Tu, Ning-Ning Zeng and Xiao-Chen Wang.* Spiro-Bicyclic Bisborane Catalysts for Metal-Free Chemoselective and Enantioselective Hydrogenation of Quinolines. Angew. Chem. Int. Ed. 2019, 58, 4664-4668.
|
|
|
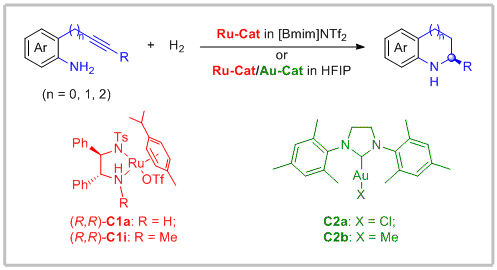
53. Cong Xu, Yu Feng, Faju Li, Jiahong Han, Yan-Mei He and Qing-Hua Fan.* A Synthetic Route to Chiral Benzo-Fused N-Heterocycles via Sequential Intramolecular Hydroamination and Asymmetric Hydrogenation of Anilino-Alkynes. Organometallics 2019, 38, 3979-3990.
|
|
|
52. Julien Pastor, Elixabete Rezabal, Arnaud Voituriez, Jean-François Betzer,* Angela Marinetti and Gilles Frison.* Revised Theoretical Model on Enantiocontrol in Phosphoric Acid Catalyzed H‑Transfer Hydrogenation of Quinoline. J. Org. Chem. 2018, 83, 2779-2787.
|

|
|
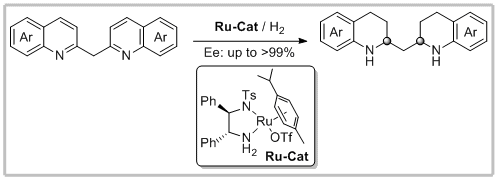
51. Bin Li, Cong Xu, Yan-Mei He,* Guo-Jun Deng and Qing-Hua Fan.* Asymmetric Hydrogenation of Bis(quinolin-2-yl)methanes: A Direct Access to Chiral 1,3-Diamines. Chin. J. Chem. 2018, 36, 1169-1173.
|
|
|
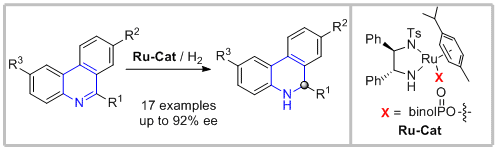
50. Zhusheng Yang, Fei Chen,* Shuxin Zhang, Yanmei He, Nianfa Yang* and Qing-Hua Fan.* A Ruthenium-Catalyzed Enantioselective Hydrogenation of Phenanthridine Derivatives. Org. Lett. 2017, 19, 1458-1461.
|
|
|
49. Xi-Chang Zhang, Yi-Hu Hu, Chuan-Fu Chen, Qiang Fang, Li-Yao Yang, Ying-Bo Lu, Lin-Jie Xie, Jing Wu,* Shijun Li* and Wenjun Fang. A Supramolecularly Tunable Chiral Diphosphine Ligand: Application to Rh and Ir-catalyzed Enantioselective Hydrogenation. Chem. Sci. 2016, 7, 4594-4599.
|

|
|
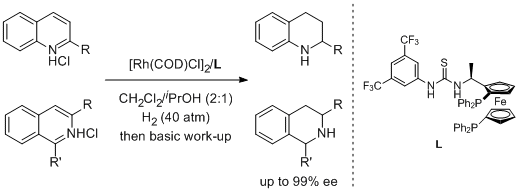
48. Jialin Wen, Renchang Tan, Shaodong Liu, Qingyang Zhao* and Xumu Zhang.* Strong Bronsted Acid Promoted Asymmetric Hydrogenation of Isoquinolines and Quinolines Catalyzed by a Rh–thiourea Chiral Phosphine Complex viaAnion Binding. Chem. Sci. 2016, 7, 3047-3051.
|
|
|
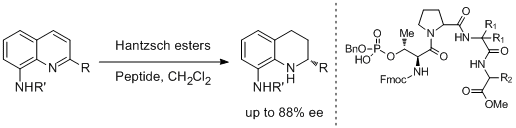
47. Christopher R. Shugrue and Scott J. Miller.* Phosphothreonine as a Catalytic Residue in Peptide-Mediated Asymmetric Transfer Hydrogenations of 8-Aminoquinolines. Angew. Chem. Int. Ed. 2015, 54, 11173-11176.
|
|
|
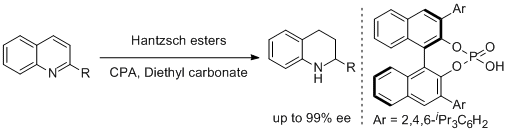
46. Ganesh V. More and Bhalchandra M. Bhanage.* Chiral Phosphoric Acid Catalyzed Asymmetric Transfer Hydrogenation of Quinolines in a Sustainable Solvent. Tetrahedron: Asymmetry 2015, 2, 1174-1179.
|
|
|
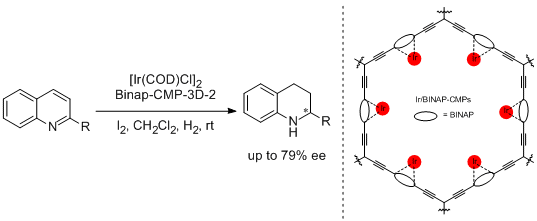
45. Xu Wang, Jun Li, Shengmei Lu, Yan Liu,* Can Li.* Efficient Enantioselective Hydrogenation of Quinolines Catalyzed by Conjugated Microporous Polymers with Embedded Chiral BINAP Ligand. Chin. J. Catal. 2015, 36, 1170-1174.
|
|
|
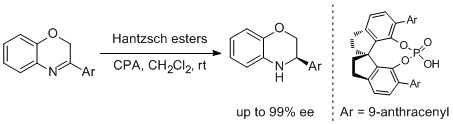
44. Yiliang Zhang, Rong Zhao, Robert Li-Yuan Bao and Lei Shi.* Highly Enantioselective SPINOL-Derived Phosphoric Acid Catalyzed Transfer Hydrogenation of Diverse C=N-Containing Heterocycles. Eur. J. Org. Chem. 2015, 3344-3351.
|
|
|
43. Joshni John, Cody Wilson-Konderka and Costa Metallinosa.* Low Pressure Asymmetric Hydrogenation of Quinolines Using an Annulated Planar ChiralN-Ferrocenyl NHC-Iridium Complex. Adv. Synth. Catal. 2015, 357, 2071-2081. |

|
|
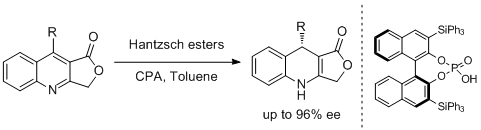
42. Alexandre Aillerie, Vincent Lemau de Talancé, Aurélien Moncomble, Till Bousquet*and Lydie Pélinski.*Enantioselective Organocatalytic Partial Transfer Hydrogenation of Lactone-Fused Quinolines. Org. Lett. 2014, 16, 2982-2985. |
|
|
41. Ferry, A., Stemper, J., Marinetti, A., Voituriez, A. and Guinchard, X. Thiophostone-Derived Bronsted Acids in the Organocatalyzed Transfer Hydrogenation of Quinolines: Influence of the P-Stereogenicity.Eur. J. Org. Chem. 2014, 188-193. |
|
|
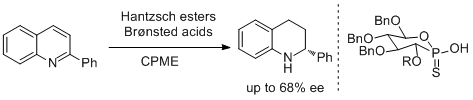
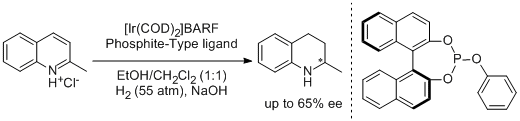
40. Sergey E. Lyubimov, Dmitry V. Ozolin, Pavel Yu Ivanov, Artem Melman, Valeriya S. Velezheva, Vadim A. Davankov. The Use of Phosphite-Type Ligands in the Ir-Catalyzed Asymmetric Hydrogenation of Heterocyclic Compounds. Chirality 2014, 26, 56-60. |
|
|
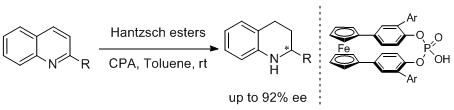
39. Jérémy Stemper, Kévin Isaac, Julien Pastor, Gilles Frison, Pascal Retailleau, Arnaud Voituriez, Jean-Francois Betzer,* and Angela Marinetti.* Development of Chiral Phosphoric Acids based on Ferrocene-Bridged Paracyclophane Frameworks. Adv. Synth. Catal. 2013, 355, 3613-3624. |
|
|
38. Zi-Yuan Ding, Tianli Wang, Yan-Mei He, Fei Chen, Hai-Feng Zhou, Qing-Hua Fan,* Qingxiang Guo,* and Albert S. C. Chan.* Highly Enantioselective Synthesis of Chiral Tetrahydroquinolines and Tetrahydro-isoquinolines by Ruthenium-Catalyzed Asymmetric Hydrogenation in Ionic Liquid. Adv. Synth. Catal. 2013, 355, 3727-3735. |

|
|
37. Xi-Feng Tu, Liu-Zhu Gong.* Highly Enantioselective Transfer Hydrogenation of Quinolines Catalyzed by Gold Phosphates: Achiral Ligand Tuning and Chiral-Anion Control of Stereoselectivity. Angew. Chem. Int. Ed. 2012, 51, 11346-11349. |
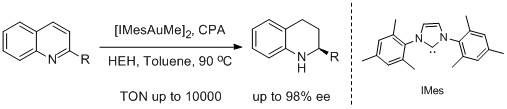
|
|
36. Anna M. Maj, Isabelle Suisse, Catherine Méliet, Christophe Hardouin and Francine Agbossou-Niedercorn.* Highly Enantioselective Hydrogenation of New 2-functionalized Quinoline Derivatives. Tetrahedron Lett. 2012, 53, 4747-4750. |

|
|
35. Tianli Wang, Lian-Gang Zhuo, Zhiwei Li, Fei Chen, Ziyuan Ding, Yanmei He, Qing-Hua Fan,* Junfeng Xiang, Zhi-Xiang Yu* and Albert S. C. Chan. Highly Enantioselective Hydrogenation of Quinolines Using Phosphine-free Chiral Cationic Ruthenium Catalysts: Scope, Mechanism, and Origin of Enantioselectivity. J. Am. Chem. Soc.2011, 133, 9878-9891. |
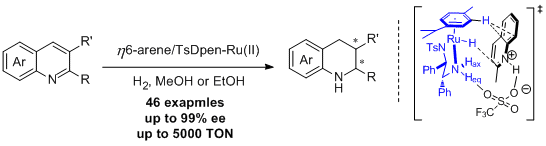
|
|
34. Magnus Rueping,* Thomas Theissmann, Mirjam Stoeckel, Andrey P. Antonchick. Direct Enantioselective Access to 4-substituted Tetrahydroquinolines by Catalytic Asymmetric Transfer Hydrogenation of Quinolines. Org. Biomol. Chem. 2011, 9, 6844-6850. |
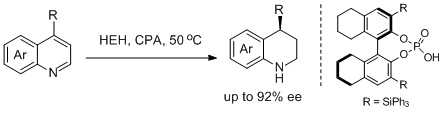
|
|
33. Tianli Wang, Guanghui Ouyang, Yan-Mei He, Qing-Hua Fan*. Asymmetric Tandem Reduction of 2-(Aroylmethyl)quinolines with Phosphine-Free Ru-TsDPEN Catalyst. Synlett 2011, 939-942. |
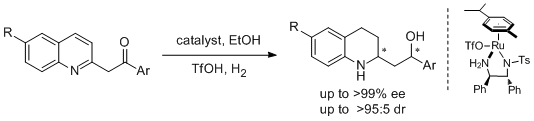
|
|
32. Magnus Rueping* and Rene M. Koenigs. Bronsted Acid Differentiated Metal Catalysis by Kinetic Discrimination. Chem. Commun. 2011, 47, 304-306. |
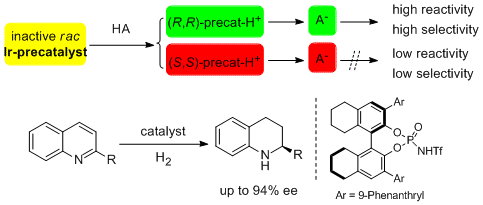
|
|
31. Jose L.Nunez-Rico, Hector Fernandez-Perez, J. Benet-Buchholz, and Anton Vidal-Ferran*, Asymmetric Hydrogenation of Heteroaromatic Compounds Mediated by Iridium-(P-OP) Complexes. Organometallics 2010, 29, 6627–6631. |
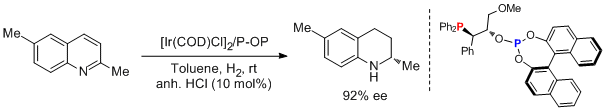
|
|
30. Wei-Jun Tang, Jing Tan, Li-Jin Xu,* Kim-Hung Lam, Qing-Hua Fan, and Albert S. C. Chan. Highly Enantioselective Hydrogenation of Quinoline and Pyridine Derivatives with Iridium-(P-Phos) Catalyst. Adv. Synth. Catal.2010, 352, 1055-1062. |
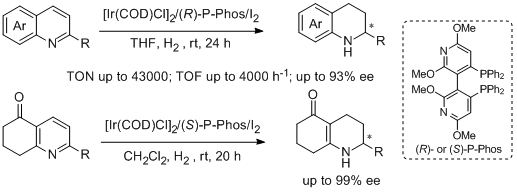
|
|
29. Fa-Rong Gou, Wei Li, Xumu Zhang,* and Yong-Min Liang.* Iridium-Catalyzed Asymmetric Hydrogenation of Quinoline Derivatives with C3*-TunePhos. Adv. Synth. Catal. 2010, 352, 2441-2444. |
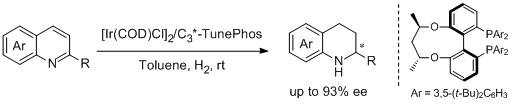
|
|
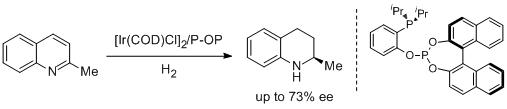
28. Miguel Rubio and Antonio Pizzano.* Application of Phosphine-Phosphite Ligands in the Iridium Catalyzed Enantioselective Hydrogenation of 2-Methylquinoline. Molecules 2010, 15, 7732-7741. |
|
|
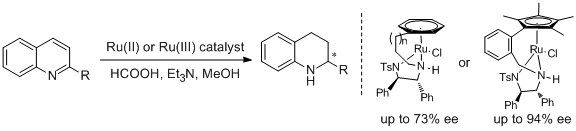
27. Vimal Parekh, James A. Ramsden, Martin Wills.* Asymmetric Transfer Hydrogenation of Quinolines Using Tethered Ru(II) Catalysts. Tetrahedron: Asymmetry 2010, 21, 1549-1556. |
|
|
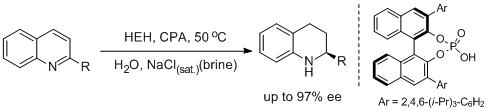
26. Magnus Rueping* and Thomas Theissmann. Asymmetric Bronsted Acid Catalysis in Aqueous Solution. Chem. Sci. 2010, 1, 473-476. |
|
|
25. Magnus Rueping,* Mirjam Stoeckel, Erli Sugiono, Thomas Theissmann. Asymmetric Metal-free Synthesis of Fluoroquinolones by Organocatalytic Hydrogenation. Tetrahedron 2010, 66, 6565-6568. |

|
|
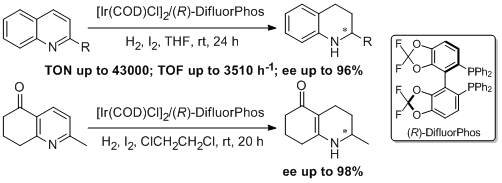
24. Weijun Tang, Yawei Sun, Lijin Xu,* Tianli Wang, Qinghua Fan, Kim-Hung Lam and Albert S. C. Chan. Highly Efficient and Enantioselective Hydrogenation of Quinolines and Pyridines with Ir-Difluorphos Catalyst. Org. Biomol. Chem. 2010, 8, 3464-3471. |
|
|
23. Maja Heitbaum, Roland Frohlich and Frank Gloriua.* Diastereoselective Hydrogenation of Substituted Quinolines to Enantiomerically Pure Decahydroquinolines. Adv. Synth. Catal. 2010, 352, 357-362. |
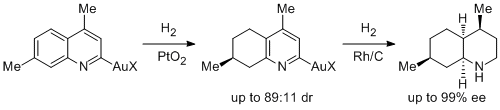
|
|
22. Zhi-Jian Wang, Hai-Feng Zhou, Tian-Li Wang, Yan-Mei He and Qing-Hua Fan.* Highly Enantioselective Hydrogenation of Quinolines under Solvent-free or Highly Concentrated Conditions. Green Chem. 2009, 11, 767-769. |
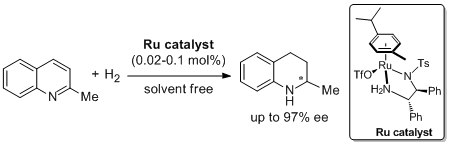
|
|
21. Hiroshi Tadaoka, Damien Cartigny, Takuto Nagano, Tushar Gosavi, Tahar Ayad, Jean-Pierre GenTt, Takashi Ohshima,* Virginie Ratovelomanana-Vidal,* and Kazushi Mashima. Unprecedented Halide Dependence on Catalytic Asymmetric Hydrogenation of 2-Aryl- and 2-Alkyl-Substituted Quinolinium Salts by Using Ir Complexes with Difluorphos and Halide Ligands. Chem. Eur. J. 2009, 15, 9990-9994. |
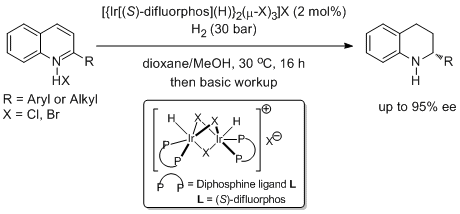
|
|
20. Chao Wang, Chaoqun Li, Xiaofeng Wu, Alan Pettman, and Jianliang Xiao.* pH-Regulated Asymmetric Transfer Hydrogenation of Quinolines in Water. Angew. Chem. Int. Ed. 2009, 48, 6524-6528. |
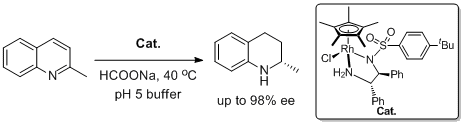
|
|
19. Matthias Eggenstein, Anika Thomas, Jens Theuerkauf, Giancarlo Francio,* and Walter Leitnera.* Highly Efficient and Versatile Phosphine-Phosphoramidite Ligands for Asymmetric Hydrogenation. Adv. Synth. Catal. 2009, 351, 725-732. |
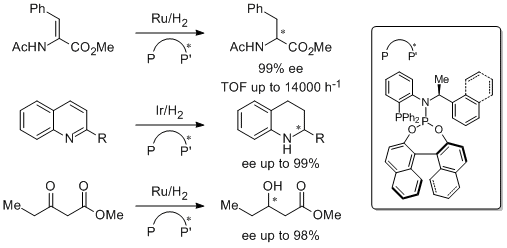
|
|
18. Metallinos Costa,* Barrett Fred B, Xu, Shufen. Bronsted Acid Catalyzed Asymmetric Reduction of 2- and 2,9-Substituted 1,10-Phenanthrolines. Synlett 2008, 720-724. |
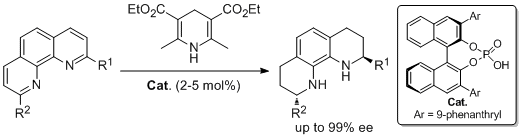
|
|
17. Haifeng Zhou, Zhiwei Li, Zhijian Wang, Tianli Wang, Lijin Xu, Yanmei He, Qing-Hua Fan,Jie Pan, Lianquan Gu, and Albert S. C. Chan.* Hydrogenation of Quinolines Using a Recyclable Phosphine-Free Chiral Cationic Ruthenium Catalyst: Enhancement of Catalyst Stability and Selectivity in an Ionic Liquid. Angew. Chem. Int. Ed. 2008, 47, 8464-8467. |
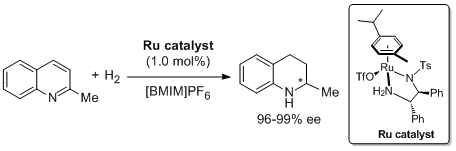
|
|
16. Natasa Mrsic, Laurent Lefort, Jeroen A. F. Boogers, Adriaan J. Minnaard,* Ben L. Feringa,* and Johannes G. de Vries.* Asymmetric Hydrogenation of Quinolines Catalyzed by Iridium Complexes of Monodentate BINOL-Derived Phosphoramidites. Adv. Synth. Catal. 2008, 350, 1081-1089. |
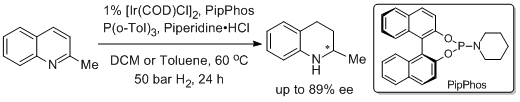
|
|
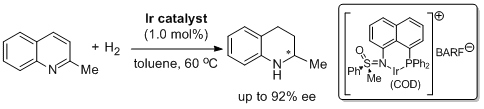
15. Sheng-Mei Lu and Carsten Bolm.* Synthesis of Sulfoximine-Derived P,N Ligands and their Applications in Asymmetric Quinoline Hydrogenations. Adv. Synth. Catal. 2008, 350, 1101-1105. |
|
|
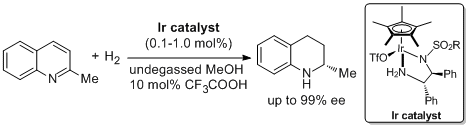
14. Zhi-Wei Li, Tian-Li Wang, Yan-Mei He, Zhi-Jian Wang, Qing-Hua Fan,* Jie Pan, and Li-Jin Xu.* Air-Stable and Phosphine-Free Iridium Catalysts for Highly Enantioselective Hydrogenation of Quinoline Derivatives. Org. Lett. 2008, 10, 5265-5268. |
|
|
13. Qun-Sheng Guo, Da-Ming Du* and Jiaxi Xu. The Development of Double Axially Chiral Phosphoric Acids and Their Catalytic Transfer Hydrogenation of Quinolines. Angew. Chem. Int. Ed. 2008, 47, 759-762. |

|
|
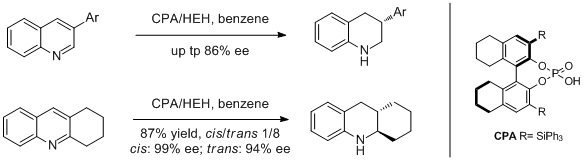
12. Magnus Rueping,* Thomas Theissmann, Sadiya Raja, and Jan W. Bats. Asymmetric Counterion Pair Catalysis: An Enantioselective Bronsted Acid-Catalyzed Protonation. Adv. Synth. Catal. 2008, 350, 1001-1006. |
|
|
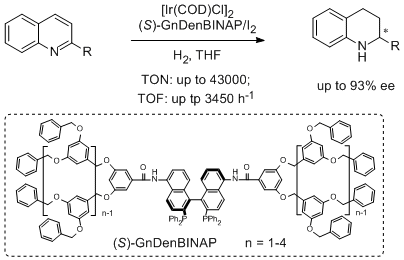
11. Zhi-Jian Wang, Guo-Jun Deng, Yong Li, Yan-Mei He, Wei-Jun Tang and Qing-Hua Fan.* Enantioselective Hydrogenation of Quinolines Catalyzed by Ir(BINAP)-Cored Dendrimers: Dramatic Enhancement of Catalytic Activity.Org. Lett. 2007, 9, 1243-1246. |
|
|
10. Coralie Deport, Marie Buchotte, Keren Abecassis, Hiroshi Tadaoka, Tahar Ayad, Takashi Ohshima, Jean-Pierre Genet,* Kazushi Mashima,* Virginie Ratovelomanana-Vidal.* Novel Ir–SYNPHOS and Ir–DIFLUORPHOS Catalysts for Asymmetric Hydrogenation of Quinolines. Synlett 2007, 2743-2747. |

|
|
9. Sau Hing Chan, Kim Hung Lam,* Yue-Ming Li, Lijin Xu, Weijun Tang, Fuk Loi Lam, Wai Hung Lo, Wing Yiu Yu, Qinghua Fan* and Albert S. C. Chan.* Asymmetric Hydrogenation of Quinolines with Recyclable and Air-stable Iridium Catalyst Systems. Tetrahedron: Asymmetry 2007, 18, 2625-2631. |
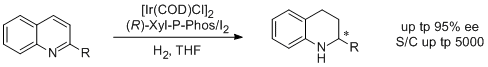
|
|
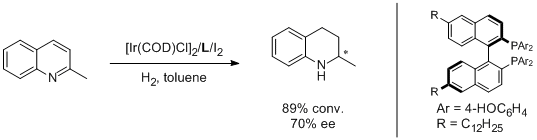
8. Mohamad Jahjah, Mohamad Alame, Stephane Pellet-Rostaing* and Marc Lemaire*. Catalytic Asymmetric Hydrogenation ofα-Ketoesters and Quinoline Uusing Electronically Enriched BINAP. Tetrahedron: Asymmetry 2007, 18, 2305-2312. |
|
|
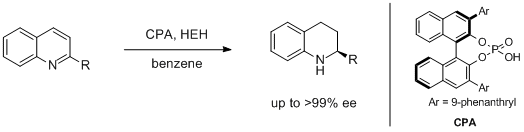
7. Wei-Jun Tang, Shou-Fei Zhu, Li-Jin Xu,* Qi-Lin Zhou, Qing-Hua Fan,* Hai-Feng Zhou, Kimhung Lam and Albert S. C. Chan.* Asymmetric Hydrogenation of Quinolines with High Substrate/Catalyst Ratio. Chem. Commun. 2007, 613-615. |
|
|
6. Magnus Rueping,* Andrey P. Antonchick, and Thomas Theissmann. A Highly Enantioselective Bronsted Acid Catalyzed Cascade Reaction: Organocatalytic Transfer Hydrogenation of Quinolines and Their Application in the Synthesis of Alkaloids. Angew. Chem. Int. Ed. 2006, 45, 3683-3686. |

|
|
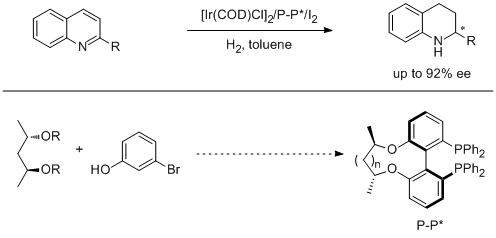
5. Liqin Qiu, Fuk Yee Kwong, Jing Wu, Wai Har Lam, Shusun Chan, Wing-Yiu Yu, Yue-Ming Li, Rongwei Guo, Zhongyuan Zhou and Albert S. C. Chan.* A New Class of Versatile Chiral-Bridged Atropisomeric Diphosphine Ligands: Remarkably Efficient Ligand Syntheses and Their Applications in Highly Enantioselective Hydrogenation Reactions. J. Am. Chem. Soc. 2006, 128, 5955-5965. |
|
|
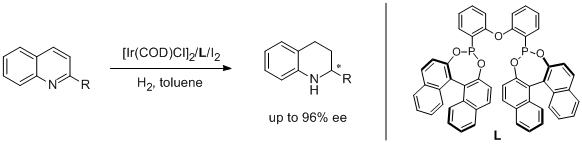
4. Manfred T. Reetz* and Xiaoguang Li. Asymmetric Hydrogenation of Quinolines Catalyzed by Iridium Complexes of BINOL-derived Diphosphonites. Chem. Commun. 2006, 2159-2160. |
|
|
3. Tsuneaki Yamagata, Hiroshi Tadaoka, Mitsuhiro Nagata, Tsukasa Hirao, Yasutaka Kataoka, Virginie Ratovelomanana-Vidal, Jean Pierre Genet* and Kazushi Mashima.* Oxidative Addition of RCO2H and HX to Chiral Diphosphine Complexes of Iridium(I): Convenient Synthesis of Mononuclear Halo-Carboxylate Iridium(III) Complexes and Cationic Dinuclear Triply Halogen-Bridged Iridium(III) Complexes and Their Catalytic Performance in Asymmetric Hydrogenation of Cyclic Imines and 2-Phenylquinoline. Organometallics 2006, 25, 2505-2513. |

|
|
2. Kim Hung Lam, Lijin Xu,* Lichun Feng, Qing-Hua Fan,* Fuk Loi Lam, Wai-hung Lo, Albert S. C. Chan.* Highly Enantioselective Iridium-Catalyzed Hydrogenation of Quinoline Derivatives Using Chiral Phosphinite H8-BINAPO. Adv. Synth. Catal. 2005, 347, 1755-1758. |
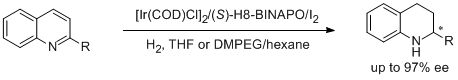
|
|
1. Lijin Xu, Kim Hung Lam, Jianxin Ji, Jing Wu, Qing-Hua Fan,* Wai-Hung Lo and Albert S. C. Chan.* Air-stable Ir-(P-Phos) complex for Highly Enantioselective Hydrogenation of Quinolines and Their Immobilization in Poly(ethylene glycol) dimethyl Ether (DMPEG). Chem. Commun. 2005, 1390-1392. |
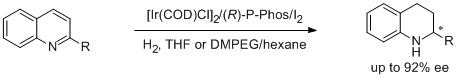
|